|
BurstDR stimulation is preferred to traditional "tingling" tonic stimulation by 87% of patients. Smallest device: Designed with daily comfort in mind6 the Eterna SCS system is the smallest implantable, rechargeable spinal cord stimulator *4 on the market.Ĭlinically proven therapy: The Eterna SCS system features Abbott's proprietary BurstDR stimulation, which mimics natural firing patterns found in the brain 7 to deliver superior **8 pain relief. With patient needs front of mind, Abbott created Eterna to be recharged less than five times a year under normal use, making it the lowest recharge burden platform on the market. The studies found that people wanted a smaller implant for comfort while reducing the need to charge the device daily or weekly. 5Ībbott developed Eterna based on extensive studies with patients, physicians and caregivers to understand the unmet needs of people living with chronic pain. *4 Eterna SCS utilizes Abbott's proprietary low-dose BurstDR™ stimulation, the only SCS waveform technology with the highest level of clinical evidence (1A evidence), proven to reduce pain 23% more than traditional waveform technology approaches. Food and Drug Administration (FDA) approval of the company's Eterna™ spinal cord stimulation (SCS) system - the smallest implantable, rechargeable spinal cord stimulator currently available on the market for the treatment of chronic pain. This neuromodulation device provides an optimized experience with the ability to wirelessly charge as few as five times per year, the lowest recharge burden compared to other rechargeable SCS systems §1,2,3ĪBBOTT PARK, IL / ACCESSWIRE / Decem/ Abbott (NYSE:ABT) today announced the U.S. 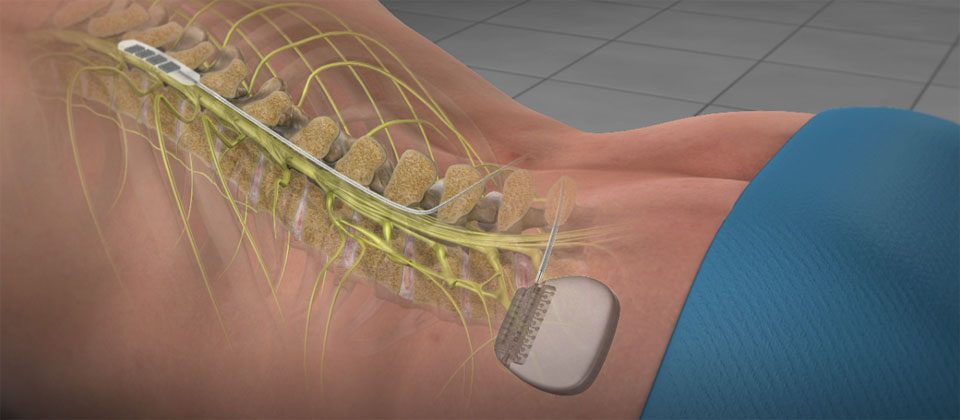
Food and Drug Administration (FDA) approved Abbott's Eterna™ spinal cord stimulation (SCS) system for the treatment of chronic pain Benzinga does not provide investment advice. MedTech Giant Abbott To Inject Over €400M In Ireland To Expand Production Of Glucose Monitoring Techīaby Formula Startup Seeks To Compete Abbott With 15% Market Share: Reportĭon't miss real-time alerts on your stocks - join Benzinga Pro for free! Try the tool that will help you invest smarter, faster, and better. Price Action: ABT shares are down 1.25% at $106.09 on the last check Tuesday. SCS therapy, also known as neurostimulation, is an implanted device that sends mild electrical pulses to the nerves along the spinal cord to relieve chronic pain.īurstDR therapy is Abbott's exclusive stimulation technology that delivers pulses – or bursts – of mild electrical energy to alter pain signals as they travel from the spinal cord to the brain. It can be used with Abbott's NeuroSphere Virtual Clinic connected care technology, which allows a person to communicate with a physician through secure in-app video chat and remotely receive stimulation settings in real-time. The Proclaim Plus SCS system is recharge-free with a battery lasting up to 10 years. The next generation of Abbott's proprietary BurstDR stimulation, FlexBurst360 therapy, offers pain coverage across up to six areas of the trunk and/or limbs and enables programming that can be adjusted as a person's therapeutic needs evolve. The FDA has approved Abbott Laboratories (NYSE: ABT) new Proclaim Plus spinal cord stimulation (SCS) system featuring FlexBurst360 therapy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
